Our research
The efficient industrial scale production of hydrogen from seawater using an electrocatalytically driven hydrogen evolution reaction (HER) would represent a game changing contributor toward the global supply of abundant green energy. Unfortunately, most conventional noble metal based catalysts for hydrogen production in pure water produce corrosive hypochlorite by-products when used in seawater, which quickly inhibit the catalytic activity and degrade the electrodes.
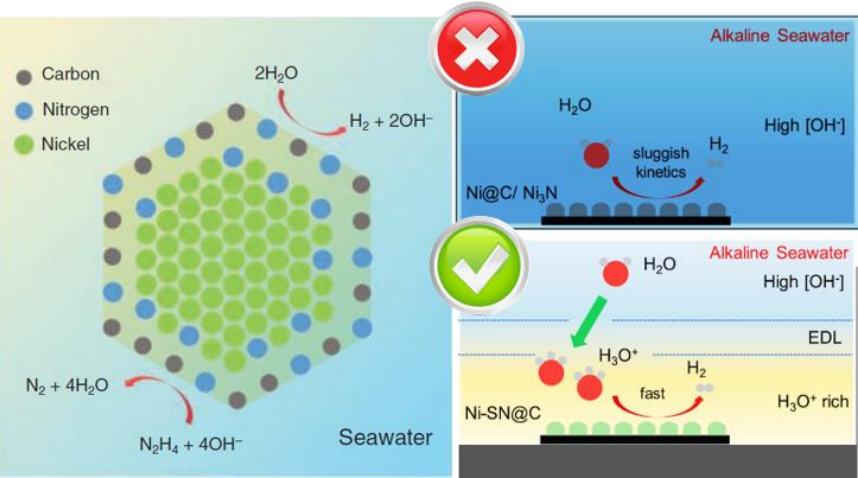
Recently, researchers from the School of Chemical Engineering and Advanced Materials at the University of Adelaide synthesised a new transition metal nanoparticle compound, Nickel surface nitride (Ni-SN@C), which demonstrated catalytic efficiency for hydrogen generation in seawater comparable or greater to that of typical platinum-carbon (Pt-C) based compounds used in pure water.
In order to optimise the catalytic material efficiency through materials engineering techniques, a detailed understanding of the surface chemistry of the catalyst is required. The researchers used the Soft X-ray beamline to investigate the surface chemistry of the Ni-SN@C nanoparticles at the atomic level. They were able to show that the nanoparticles consisted of a pure metallic Nickel core, with a surface layer of nickel nitride. Importantly, this nickel-nitride layer exhibited reactive dangling bonds which are thought to be responsible for the increased catalytic activity compared to bulk Nickel Nitride (Ni3N).